Description
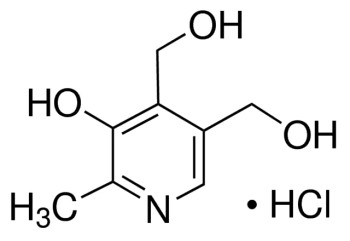
Glucosamine hydrochloride
Pharmaceutical Secondary Standard; Certified Reference Material
Synonym: 2-Amino-2-deoxy-D-glucose hydrochloride, D-(+)-Glucosamine hydrochloride, Chitosamine hydrochloride
-
CAS Number 66-84-2
-
Empirical Formula (Hill Notation) C6H13NO5 · HCl
-
Molecular Weight 215.63
-
Beilstein/REAXYS Number 4157370
-
MDL number MFCD00135831
-
PubChem Substance ID 329823206
-
NACRES NA.24
General description
Pharmaceutical secondary standards for application in quality control, provide pharma laboratories and manufacturers with a convenient and cost-effective alternative to the preparation of in-house working standards.
Glucosamine hydrochloride is a drug prescribed for the remediation of degenerative joint diseases in small animals.[1]
Application
Glucosamine hydrochloride may be used as a pharmaceutical reference standard for the determination of the analyte in pharmaceutical formulations and plasma samples by high performance liquid chromatography (HPLC).[1][2][3]
These Secondary Standards are qualified as Certified Reference Materials. These are suitable for use in several analytical applications including but not limited to pharma release testing, pharma method development for qualitative and quantitative analyses, food and beverage quality control testing, and other calibration requirements.
Biochem/physiol Actions
Glucosamine, an amino sugar, is the precursor of the hexosamine biosynthetic pathway leading to the formation of UDP-N-acetylglucosamine (UDP-GlcNAc), which is then used for making glycosaminoglycans, proteoglycans, and glycolipids. D-(+)-Glucosamine inhibits the coaggregation of Candida yeast species with the bacterial strain S. salivarius.
Analysis Note
These secondary standards offer multi-traceability to the USP, EP (PhEur) and BP primary standards, where they are available.
Other Notes
This Certified Reference Material (CRM) is produced and certified in accordance with ISO 17034 and ISO/IEC 17025. All information regarding the use of this CRM can be found on the certificate of analysis.
Footnote
To see an example of a Certificate of Analysis for this material enter LRAB3705 in the slot below. This is an example certificate only and may not be the lot that you receive.
| Related Categories | Analytical Standards, Analytical/Chromatography, E-J, Pharmaceutical Secondary Standards, Pharmacopeia & Metrological Institutes Standards Less... |
| Quality Level | 300 |
| grade | certified reference material |
| - | pharmaceutical secondary standard |
| form | neat |
| mp | 190-194 °C (dec.) (lit.) |
| Featured Industry | Pharmaceutical (small molecule) |
| format | neat |
| pharmacopeia traceability | traceable to PhEur Y0001406 |
| - | traceable to USP 1294207 |
| SMILES string | Cl.N[C@H]1C(O)O[C@H](CO)[C@@H](O)[C@@H]1O |
| InChI | 1S/C6H13NO5.ClH/c7-3-5(10)4(9)2(1-8)12-6(3)11;/h2-6,8-11H,1,7H2;1H/t2-,3-,4-,5-,6?;/m1./s1 |
| InChI key | QKPLRMLTKYXDST-NSEZLWDYSA-N |