Description
Tartaric Acid, 1G, Pharmaceutical Secondary Standard; Certified Reference Material
Synonym(s):
L-(+)-Tartaric acid, L-Threaric acid, (2R,3R)-(+)-Tartaric acid
Linear Formula:
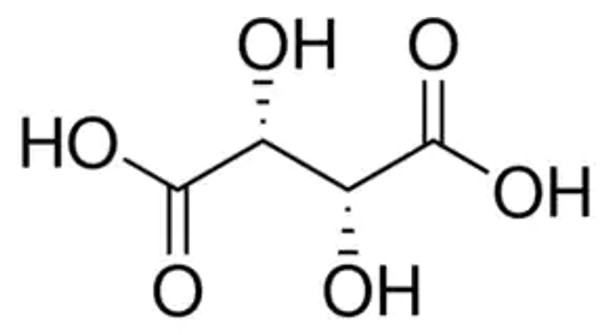
HO2CCH(OH)CH(OH)CO2H
CAS Number:
87-69-4
Molecular Weight:
150.09
Beilstein:
1725147
EC Number:
201-766-0
MDL number:
MFCD00064207
PubChem Substance ID:
329823401
NACRES:
NA.24
PROPERTIES
Quality Level
300
grade
certified reference material
pharmaceutical secondary standard
Agency
traceable to USP 1643340
vapor density
5.18 (vs air)
CofA
current certificate can be downloaded
autoignition temp.
797 °F
packaging
pkg of 1 g
technique(s)
HPLC: suitable
gas chromatography (GC): suitable
mp
170-172 °C (lit.)
application(s)
pharmaceutical (small molecule)
format
neat
storage temp.
2-30°C
SMILES string
O[C@H]([C@@H](O)C(O)=O)C(O)=O
InChI
1S/C4H6O6/c5-1(3(7)8)2(6)4(9)10/h1-2,5-6H,(H,7,8)(H,9,10)/t1-,2-/m1/s1
InChI key
FEWJPZIEWOKRBE-JCYAYHJZSA-N
General description
Tartaric Acid belongs to the group of carboxylic acids, and is abundantly found in grapes and wine. It is widely used in drugs, food, and beverage industry.
Certified pharmaceutical secondary standards for application in quality control provide pharma laboratories and manufacturers with a convenient and cost-effective alternative to pharmacopeia primary standards
Application
These Secondary Standards are qualified as Certified Reference Materials. These are suitable for use in several analytical applications including but not limited to pharma release testing, pharma method development for qualitative and quantitative analyses, food and beverage quality control testing, and other calibration requirements.
Tartaric Acid may be used as a pharmaceutical reference standard for the quantification of the analyte in pharmaceutical formulations using stopped-flow chemiluminescence spectrometry.
Analysis Note
These secondary standards offer multi-traceability to the USP, EP and BP primary standards, where they are available.
Other Notes
This Certified Reference Material (CRM) is produced and certified in accordance with ISO 17034 and ISO/IEC 17025. All information regarding the use of this CRM can be found on the certificate of analysis.
SAFETY INFORMATION
Signal Word
Danger
Hazard Statements
H318
Precautionary Statements
P280 - P305 + P351 + P338
Hazard Classifications
Eye Dam. 1
Storage Class Code
11 - Combustible Solids
WGK
WGK 1
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable